
Proton-deficient nuclei undergo beta decay - emitting a beta particle (electron) and an antineutrino to convert a neutron to a proton - thus raising the elements atomic number Z by one. Proton-deficient or neutron-deficient nuclei undergo nuclear decay reactions that serve to correct unbalanced neutron/proton ratios. Other heavy unstable elements undergo fission reactions in which they split into nuclei of about equal size. 1) An electron from the closest energy level falls into the nucleus, which causes a proton to become a neutron. Alpha decay is a form of spontaneous fission, a reaction in which a massive nuclei can lower its mass and atomic number by splitting. A positron decay equation: -> Five more, but no answers. decay, or positron emission, and electron capture. The energy released in an alpha decay reaction is mostly carried away by the lighter helium, with a small amount of energy manifesting itself in the recoil of the much heavier daughter nucleus. Get Britannica Premium for only 24.95 - a 67 discount Subscribe Now. Therefore, the mass of the parent atom must simply be greater than the sum of the masses of its daughter atom and the helium atom. Since the number of total protons on each side of the reaction does not change, equal numbers of electrons are added to each side to make neutral atoms. Electron capture is when an atomic electron is absorbed by a proton in. Both electron capture and electron-proton collisions have the same decay equation. However, when they interact with each other, it is the weak interaction that facilitates the collision. Isot.\( \newcommand\]Īs with beta decay and electron capture, Δm must only be less than zero for spontaneous alpha decay to occur. Electrons and protons are attracted to each other via the electromagnetic interaction. Skin Dose: Point Source: Disk Source: Reported for 1 Ci over 10 cm2 of skin 40.5 mrad/hr (gamma dose) 0 mrad/hr (beta dose) 0 mrad/hr (beta dose) Detection Information: Usable Detectors listed with estimated efficiencies (Use efficiencies listed on instrument when available) Ludlum 3 with pancake probe at 1 cm: <1 Liq. C 41, 030003 (2017)Ĭharged particle cross section database for medical radioisotope production. Qtool: Calculation of Reaction Q-values and thresholds, Los Alamos National Laboratory, T-2 Nuclear Information Service. NuDat 3.0 software, National Nuclear Data Center, Brookhaven National Laboratory, Available from: IAEA-TECDOC-1863, Gallium-68 cyclotron production, Vienna, 2019 IAEA-TECDOC-2, Production of long-lived parent radionuclides for generators: 68Ge, 82Sr, 90Sr and 188W, Vienna, 2010 IAEA-TECDOC-468, Cyclotron Produced Radionuclides: Physical Characteristics and Production Methods, Vienna, 2009 Electron Capture Decay with gamma ray emission. Cross section data of the present work are found to be in good agreement with the literature data within experimental uncertainties for 68Ge, 66Ga and 65Zn while there are significant differences for 67Ga and 69Ge. The atomic number of the parent is reduced by 1 in this process. The experimentally measured cross section data were compared with the earlier published literature data and theoretical data from TENDL-2021 nuclear data library based on the TALYS-1.96 code. 
Using the experimentally measured cross section data and stopping power of natural Zn, thick target yields were calculated for these medicinally important radionuclides. Q: Based on nuclear stability, what is the symbol for the most likely product nuclide when cesium-129. When the nuclide gallium-67 decays by electron capture. Activation cross sections of long-lived radioisotopes of 68,69Ge, 66,67Ga and 65Zn produced from alpha irradiation of natural Zn targets were measured from their respective threshold up to 46.3 MeV. Complete the nuclear chemical equation below so. The decay of Ga 67 has been investigated with scintillation spectrometers, single and in coincidence. Production and radiochemical separation of a particular radioisotope from natural zinc target with minimum contamination from other isotopes require accurate activation cross section data of all the produced radionuclides. So 68Ga can be produced for its practical application in medicine using 68Ge/ 68Ga generator where in-house medical cyclotron facilities are unavailable. They have different applications in diagnosis and therapy in nuclear medicine depending on their decay properties and half-lives. The scheme of Ga-67 decay and its energetic characteristics are taken from Nudat 2.5. 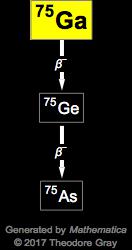
Three medicinally important radioisotopes of Ga, 66,67,68Ga, having half-lives ranging from minutes to days, could be produced from natZn(α,x) reactions. with the half-life T 1/2 78.3 hrs decays to stable 67 Zn by electron capture.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
